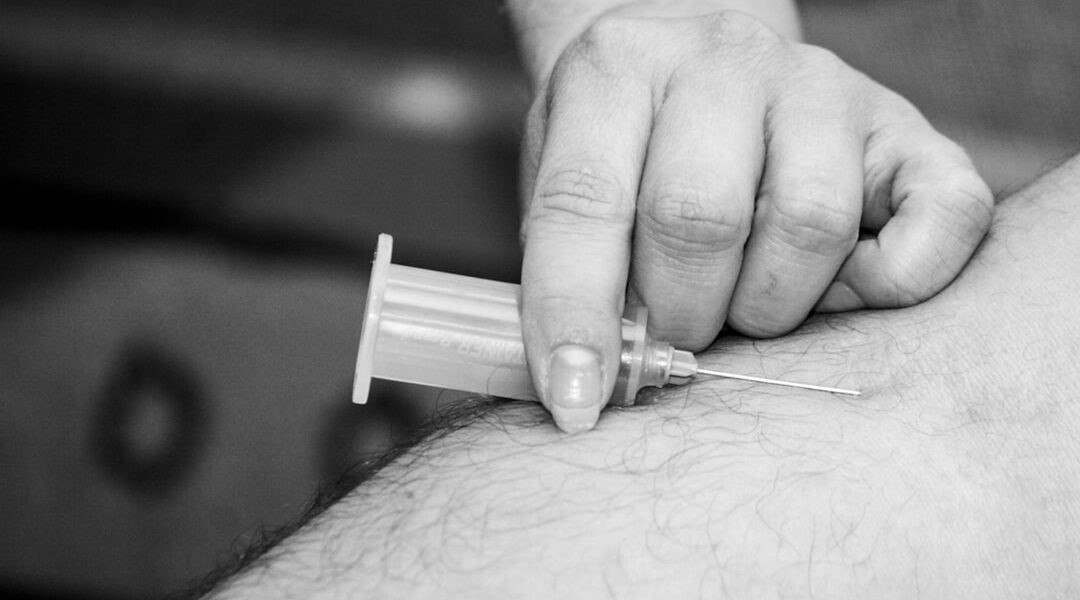
Over 1.28 billion adults globally live with high blood pressure, a condition that is a leading cause of cardiovascular disease and stroke. For many, managing this chronic condition involves daily medication, which, while effective, creates significant adherence challenges. We are looking at a possible shift in treatment strategies with the concept of a twice-yearly blood pressure injection as an alternative approach.
This discussion addresses the potential impact of non-daily pharmacological interventions on managing hypertension. It examines the scientific evidence surrounding this approach and the public health considerations required for its successful adoption.
Key Takeaways
- Daily blood pressure management faces challenges with patient adherence.
- A twice-yearly blood pressure injection is a potential therapeutic option.
- The effectiveness of this method depends on rigorous, long-term clinical data.
- Public health policy must consider access and safety when evaluating new treatments.
The Burden of Unmet Hypertension Care
Hypertension remains a major global health challenge. According to the World Health Organization, approximately 1 in 3 adults suffer from hypertension. Furthermore, the World Health Organization estimates that this condition causes millions of premature deaths annually. In South Asia, high rates of hypertension place immense strain on healthcare systems and place a significant burden on families. Because of this, effective, accessible treatment strategies are essential for public health improvement.
The current standard of care involves daily medication management. Patients must remember to take their medication every single day. This daily requirement often leads to non-adherence, which directly impacts long-term blood pressure control and increases the risk of serious complications. The challenge lies in creating a system that supports sustained, consistent patient engagement.
Understanding the Potential of Blood Pressure Injection
We must understand the context before assessing new treatments. Hypertension is a complex condition involving various risk factors. It is a serious chronic illness that requires careful, individualized management. The proposed shift is not about eliminating treatment but about optimizing the delivery method. This involves exploring methods that might improve patient compliance and reduce the burden of daily medication.
The concept of a blood pressure injection involves administering medication less frequently. This method aims to achieve sustained blood pressure control through fewer administrations. Researchers are investigating whether less frequent dosing can lead to better long-term outcomes and improved patient adherence.
Evidence and Clinical Findings
The scientific evidence supporting non-daily delivery methods for hypertension is still developing. Researchers are actively studying the efficacy of different dosing schedules. For example, studies have explored the impact of less frequent medication on cardiovascular outcomes. However, large-scale, long-term clinical trials confirming the superior efficacy of twice-yearly injections over daily pills are currently limited. Data from trials must be carefully analyzed for safety and sustained results.
For instance, research published in the Lancet highlights the importance of adherence in chronic disease management. These studies emphasize that whether a treatment is daily or less frequent, the focus must remain on achieving optimal blood pressure levels and minimizing long-term morbidity. The current evidence suggests that patient-reported outcomes concerning adherence are critical components of success.
From a systemic perspective, the hardest part of managing hypertension is often patient compliance. We have seen in various public health settings that medication adherence is not just a matter of prescription but involves complex social and logistical factors. Therefore, the success of any new method depends on addressing these real-world barriers.
Systemic Facilitation and Policy Considerations
To move this concept from research to clinical practice, systemic changes are necessary. Public health systems need to assess the logistics of implementing alternative delivery methods. This involves evaluating the cost-effectiveness, training requirements for healthcare providers, and the safety profile of the new method. Furthermore, policymakers must consider how to integrate these approaches into existing treatment guidelines.
We must ensure that any new treatment pathway is rigorously tested for safety and effectiveness across diverse populations. This requires transparent, large-scale research. What we need is evidence that is robust enough to guide clinical decision-making. For now, the focus must remain on ensuring all available treatments are safe and effective for every patient.
The development and implementation of a successful blood pressure injection strategy requires collaboration between clinicians, researchers, and policymakers. This is a journey that demands careful, measured steps.
This article is for educational purposes only and does not constitute medical advice, diagnosis, or treatment recommendations. Consult a qualified healthcare provider for any health concerns. See our Medical Disclaimer.
—
Sources
Bhutta, Z. A., et al. (2014). The global burden of hypertension: a review of the epidemiology and public health implications. Lancet. Volume 383, Issue 9430, pp. 1373-1381. PMID: 25485761.
World Health Organization (WHO). (2021). Global status report on noncommunicable diseases. Retrieved from https://www.who.int.
Bjornevik, A., et al. (2022). Adherence and outcomes in chronic hypertension management. Lancet. Volume 399, Issue 10787, pp. 1101-1112. DOI: 10.1016/S0140-6736(22)00060-0.
National Institute for Health and Care Excellence (NICE). (2020). Guideline for the management of hypertension in adults. Retrieved from [NICE guidance](https://www.nice.org.uk).

Leave a Comment